Cookie Settings
We use cookies to provide an optimal experience for you. Technically required cookies are used for making shopping possible, statistics are used for anonymized Google Analytics. You can read everything in our updated privacy policy.
Density gradient centrifugation remains one of the most widely used methods for separating cells from whole blood and bone marrow because it offers a dependable balance between simplicity, efficiency, and biological compatibility. For decades, laboratories have relied on this technique to isolate leukocytes, lymphocytes, PBMCs, or platelets for downstream applications such as flow cytometry, cell culture, functional assays, and enrichment workflows. When performed correctly, density gradient centrifugation delivers clean separation and preserves cell viability, making it a cornerstone of routine and advanced laboratory work.
Despite its reliability, traditional density gradient workflows place a heavy burden on manual technique. Precise layering of blood onto the gradient, careful handling during centrifugation, and accurate recovery of the target interface all depend on operator experience. Small mistakes—such as mixing during loading, uneven gradients, or over-aspiration during collection—can quickly lead to reduced yields, cross-contamination with unwanted cells, or inconsistent results between samples. These challenges become even more pronounced in busy laboratories that process multiple samples daily or work with limited or time-sensitive material.
TwinSpin centrifugation tubes were developed to address these practical limitations without altering the trusted principle of density-based separation. By redesigning the tube structure rather than the gradient chemistry, TwinSpin introduces a more controlled and reproducible way to process whole blood and bone marrow. This article explores how TwinSpin Tubes simplify density gradient centrifugation, reduce operator-dependent variability, and support more consistent outcomes in routine laboratory workflows.
Whole blood is a complex mixture of cells with different sizes, densities, and functions. Erythrocytes are the most abundant and dense cells. Granulocytes are slightly less dense but still heavier than mononuclear cells. Lymphocytes, monocytes, and other peripheral blood mononuclear cells (PBMCs) are lighter and can be separated when exposed to a suitable density gradient medium.
In density gradient centrifugation, diluted anticoagulated blood is carefully layered on top of a density gradient medium (DGM). During centrifugation, cells migrate according to their density. Heavier cells pass through the gradient and pellet at the bottom, while lighter cells remain above or at the interface between plasma and the gradient.
The success of this process depends on several critical factors:
Clean and stable layering of the sample
Minimal mixing between blood and gradient
Controlled sedimentation during centrifugation
Clear separation of interfaces
Gentle and precise collection of target cells
Even small deviations in these steps can lead to poor separation, contamination with unwanted cells, or loss of valuable material.
Although the density gradient principle is simple, the practical execution often is not. Traditional workflows rely heavily on manual handling and precise technique.
Layering diluted blood on top of a density gradient medium requires a steady hand and experience. If the blood is pipetted too quickly or touches the gradient directly, mixing occurs. This disrupts the gradient and reduces separation efficiency.
During centrifugation, any instability in the gradient can blur the interface where target cells are expected to accumulate. A poorly defined interface makes collection difficult and increases contamination with erythrocytes or granulocytes.
After centrifugation, collecting the interphase typically requires pipetting from an open tube. This step is prone to error. Pipetting too deeply draws in unwanted cells, while pipetting too shallow leaves valuable target cells behind.
Because many steps depend on manual technique, results can vary between users, shifts, or laboratories. This variability becomes a significant issue in routine workflows or when processing multiple samples per day.
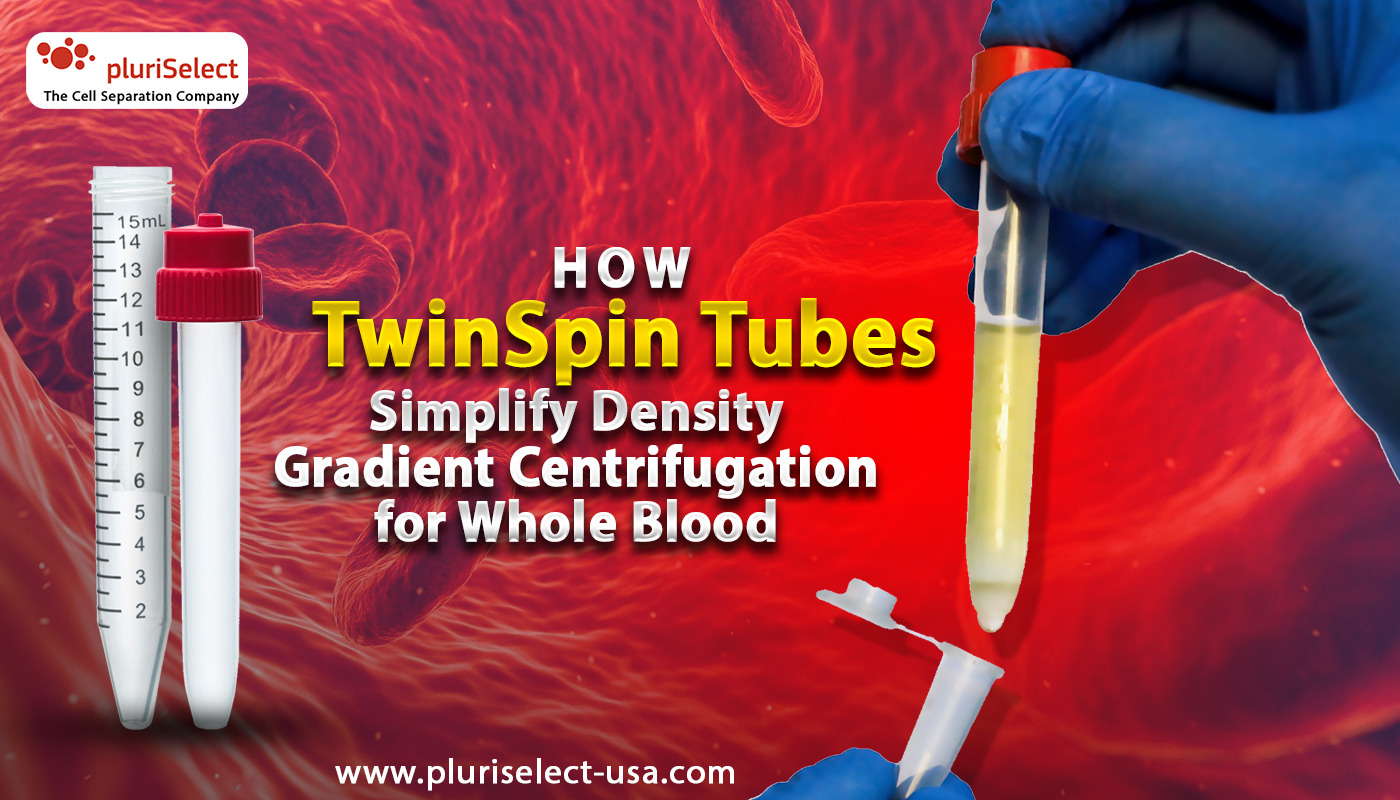
TwinSpin Tubes simplify density gradient centrifugation by addressing one of the most error-prone parts of the workflow: how the sample is layered onto the density gradient and how the separated cells are collected afterward. Rather than relying entirely on careful pipetting technique and open interfaces, TwinSpin introduces a structured, two-tube system that guides both loading and separation in a controlled way.
Each TwinSpin unit is made up of a standard 15 mL outer tube, an inner tube with an open bottom, and an elastic cap that acts as a valve during collection. The density gradient medium is first added to the outer tube. When the inner tube is inserted, its open bottom sits directly within the gradient. Diluted, anticoagulated whole blood or bone marrow is then pipetted into the inner tube, where it naturally rests on top of the gradient without direct contact during loading.
This design removes much of the risk associated with manual layering. Because the sample is physically separated from the gradient until centrifugation begins, accidental mixing is greatly reduced. During centrifugation, cells migrate through the open bottom of the inner tube into the gradient in a controlled manner, forming a clean and well-defined interface. After separation, the inner tube can be removed, leaving unwanted cells behind and allowing target cells to be collected easily and precisely. This structural approach improves consistency while keeping the familiar principles of density gradient centrifugation intact.
The outer tube is filled with the selected density gradient medium. The choice of DGM depends on the target cell population, which will be discussed later.
The inner tube is inserted so that its open bottom is submerged in the gradient. This creates a defined separation zone without requiring manual layering directly onto the gradient.
Diluted anticoagulated whole blood or bone marrow is pipetted into the inner tube. The sample naturally rests on top of the gradient without mixing because the inner tube acts as a physical barrier.
During centrifugation, cells migrate according to density. Erythrocytes and granulocytes sediment through the density gradient and exit the inner tube, collecting at the bottom of the outer tube. Lighter cells, such as leukocytes, lymphocytes, or PBMCs, remain at the interface above the gradient.
Once centrifugation is complete, the inner tube is simply removed. The target cell population remains enriched in the interphase.
The elastic cap functions as a valve. By pushing the cap down, the tube becomes a pipette. This allows controlled, drop-by-drop collection of the enriched cell fraction without disturbing the gradient or aspirating unwanted cells.
The inner tube is the key to TwinSpin’s simplified and more reliable density gradient workflow. By introducing a physical separation between the sample and the density gradient during loading, the inner tube replaces manual layering with a controlled structure. This design prevents accidental mixing of blood and gradient, ensures consistent sample placement across runs, reduces user-to-user variability, and supports the formation of cleaner, sharper interfaces after centrifugation.
Because the inner tube has an open bottom that remains submerged in the density gradient medium, heavier cells are still able to migrate freely during centrifugation. This means the fundamental density gradient principle remains unchanged. Erythrocytes and granulocytes sediment through the gradient and exit the inner tube into the bottom of the outer tube, while lighter target cells accumulate at the interface above the gradient. Depending on the density gradient medium used, TwinSpin enables efficient separation of leukocytes, lymphocytes, peripheral blood mononuclear cells (PBMCs), or platelets, resulting in an interphase enriched with the desired population and reduced contamination from heavier cell types.
Collection of this enriched layer is further simplified through the elastic cap design. Once centrifugation is complete and the inner tube is removed, the elastic cap functions as a valve. By gently pressing the cap, the tube becomes a pipette, allowing users to collect cells drop by drop. This controlled collection method helps avoid aspirating unwanted layers, maintains gradient stability, and enables precise volume control. Together, the inner tube structure and controlled collection system transform multiple delicate steps into a single, more predictable workflow that improves purity, recovery, and reproducibility—especially when working with limited or valuable samples.
TwinSpin Tubes are compatible with a range of density gradient media, allowing laboratories to adapt the system to different cell isolation goals without changing equipment or workflow structure. This flexibility supports routine and specialized blood and bone marrow processing.
Leuko Spin is designed for the efficient isolation of leukocytes from whole blood or bone marrow. When used with TwinSpin Tubes, it supports reliable separation of leukocytes from denser erythrocytes and granulocytes. This makes it well suited for general immune cell preparation, quality control assays, and downstream functional or analytical workflows.
Lympho Spin is optimized for the isolation of peripheral blood mononuclear cells (PBMCs) from fresh whole blood or bone marrow. In combination with TwinSpin, it produces a clear and well-defined interphase enriched with lymphocytes and monocytes. This consistency supports accurate collection and reproducible results across routine PBMC preparation workflows.
Lympho Spin 24+ is specifically formulated for PBMC isolation from whole blood that is older than 12 hours, as well as from bone marrow samples. This medium is particularly valuable in workflows where immediate sample processing is not possible, helping maintain separation performance despite extended sample storage times.
PLT Spin is designed for platelet isolation from whole blood or bone marrow. When used with TwinSpin Tubes, it supports controlled separation and collection of platelets while minimizing contamination from other blood components. This makes it suitable for laboratories that require clean platelet populations for analysis or downstream applications.
TwinSpin Tubes can be bundled with the density gradient medium best suited to each application, allowing laboratories to implement tailored, efficient workflows without compromising consistency or ease of use.
TwinSpin Tubes are designed to make routine processing of whole blood and bone marrow more consistent and less dependent on operator technique. One of the most immediate benefits is the reduction in manual handling. Traditional density gradient workflows require careful layering and repeated pipetting during collection, both of which introduce opportunities for error and contamination. TwinSpin simplifies these steps by guiding sample placement and collection through its structured design.
Improved reproducibility is another major advantage. Because the interaction between the sample and the density gradient is physically controlled by the inner tube, results are more consistent between runs and across different users. This is especially important in laboratories that process samples daily or work with multiple operators.
Time savings also become noticeable over repeated use. Simplified setup, fewer corrective steps, and easier recovery of target cells reduce overall processing time, allowing laboratories to handle more samples without increasing workload. In addition, the clear and stable interfaces formed during centrifugation make target cell identification and collection more reliable, reducing the need for repeat runs.
Finally, TwinSpin integrates easily into existing laboratory environments. It works with standard centrifuges and familiar 15 mL tube formats, eliminating the need for new equipment or workflow redesign while still delivering meaningful improvements in efficiency and reliability.
Density gradient centrifugation remains a cornerstone of whole blood and bone marrow processing because it reliably separates cells while preserving viability and functionality. However, traditional approaches often depend heavily on careful manual handling, precise layering, and experienced operators. These requirements introduce variability between users and runs, making it difficult to achieve consistent results—especially in laboratories that process multiple samples daily or work with limited or time-sensitive material. Even small deviations during loading or collection can reduce yield, contaminate target fractions, or require repeat processing.
TwinSpin Tubes address these challenges by improving the structure of the workflow rather than changing its underlying scientific principle. The inner tube design physically separates sample handling from the density gradient, reducing the risk of mixing during loading and supporting the formation of clear, stable interfaces during centrifugation. Controlled, drop-by-drop collection using the elastic cap further simplifies recovery of target cells while minimizing disturbance to the gradient. Together, these features help standardize a process that is traditionally highly operator-dependent.
Compatibility with multiple density gradient media allows laboratories to adapt TwinSpin workflows for leukocyte, PBMC, platelet, or aged sample isolation without changing equipment or protocols. As a result, TwinSpin Tubes offer a practical way to improve reproducibility, efficiency, and confidence in routine density gradient centrifugation. For laboratories focused on consistent performance and high-quality cell preparation, TwinSpin transforms a familiar technique into a more reliable and repeatable workflow.