Cookie Settings
We use cookies to provide an optimal experience for you. Technically required cookies are used for making shopping possible, statistics are used for anonymized Google Analytics. You can read everything in our updated privacy policy.
In cell isolation workflows, the quality of the starting material often determines the success of every step that follows. Whether the goal is to isolate leukocytes, enrich peripheral blood mononuclear cells (PBMCs), or recover rare immune populations, working with a sample that already contains a high concentration of target cells can dramatically improve efficiency and reproducibility. This is where the buffy coat plays a central role.
A buffy coat, also known as a leukocyte concentrate, is a fraction derived from whole blood during routine blood donation processing. While its original purpose is to remove leukocytes from transfusion products to improve safety and shelf life, this same process creates a highly valuable byproduct for research and laboratory use. Compared to whole blood, buffy coat contains leukocytes at concentrations that are 10 to 20 times higher, making it an ideal starting point for cell isolation workflows.
For laboratories working with immune cells, buffy coat offers several practical advantages. High cell numbers can be accessed quickly, often without extensive sample preparation. Density gradient centrifugation becomes more efficient, interfaces are easier to identify, and downstream enrichment steps benefit from reduced background cells. These characteristics are especially important when isolating rare cell types or when multiple target populations need to be recovered from a single sample.
This article provides a practical guide to understanding what a buffy coat is, how it is generated, and why it remains such a powerful resource for cell isolation workflows. By exploring both the biological and procedural aspects of buffy coat preparation and use, it aims to help laboratories make informed decisions and build more efficient, reliable cell separation strategies.
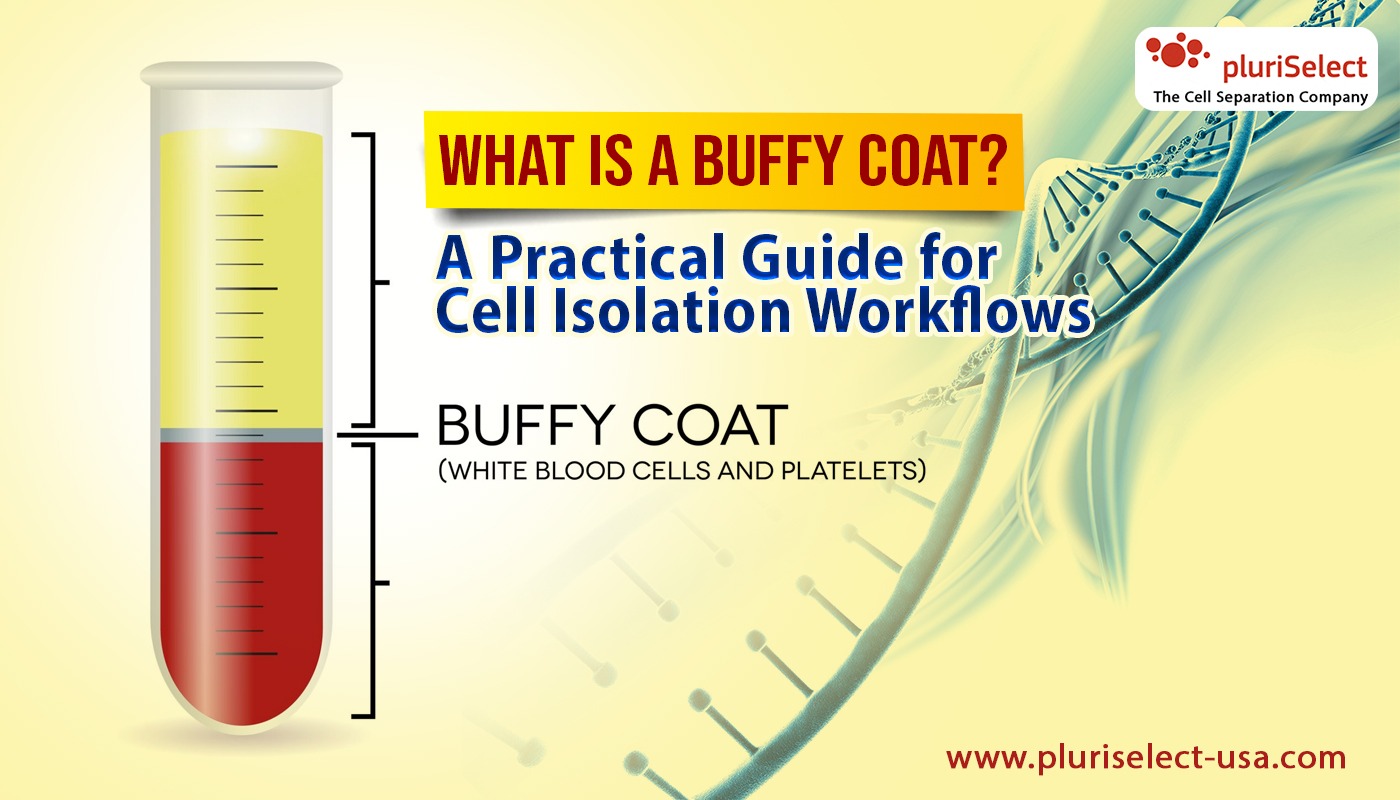
A buffy coat is a concentrated layer of white blood cells that forms when whole blood is separated into its main components. It sits between the red blood cell layer at the bottom and the plasma layer at the top after centrifugation or filtration. Because this layer is rich in leukocytes, it is often referred to as a leukocyte concentrate.
Buffy coat can be obtained in several ways. Blood banks generate it during standard donation processing, making it readily available in controlled and well-documented formats. Laboratories can also prepare buffy coat from fresh whole blood using low-speed centrifugation, physically separating the leukocyte-rich interphase without chemical additives or polymer solutions.
Buffy coat is produced when whole blood is physically separated into its main components. This separation relies on differences in cell size and density and does not require chemical additives or polymer solutions. Because of this, buffy coat preparation is considered a gentle and reliable process that preserves cell integrity.
There are two primary ways buffy coat is generated: through blood donation processing and through laboratory preparation from fresh whole blood.
In blood banks and donation centers, whole blood is routinely processed to create transfusion products. The goal is to separate red blood cells, plasma, and platelets while removing leukocytes to improve compatibility and shelf life.
The process typically follows these steps:
Whole blood is collected into anticoagulant-treated blood bags
The blood unit is centrifuged under controlled conditions
Centrifugation separates the blood into three visible layers:
Red blood cells at the bottom
Buffy coat in the middle
Plasma at the top
Key characteristics of donation-derived buffy coat:
Leukocytes are concentrated 10–20 times compared to whole blood
Red blood cells and most platelets are already removed
The material is collected as a defined fraction
No polymer-based separation media are used
Because transfusion-grade blood must contain very low leukocyte levels, the buffy coat becomes a byproduct. For research laboratories, this byproduct is a highly valuable source of immune cells.
An alternative approach used in blood processing is leukocyte filtration.
In this method:
Whole blood is passed through a leukocyte filter
Leukocytes are physically retained by the filter
Red blood cells and plasma pass through
The retained leukocytes can then be recovered as a leukocyte-rich fraction. This approach also produces a buffy coat–like material suitable for downstream cell isolation, though recovery methods may vary depending on filter design.
Laboratories can also generate buffy coat directly from fresh whole blood using a simple centrifugation protocol. This approach is useful when donation-derived buffy coat is not available.
A common lab-scale method includes:
Mixing one part whole blood with one part washing buffer
Centrifuging the diluted blood for 10 minutes at 200 × g
Using no brake to avoid disturbing layers
Identifying and removing the leukocyte-rich interphase
Advantages of in-lab preparation:
No special reagents required
Physical separation only
Compatible with downstream density centrifugation
Suitable for small sample volumes
Regardless of the method used, the goal of buffy coat generation is the same:
Reduce red blood cell background
Enrich leukocytes
Improve efficiency of downstream cell separation
By starting with buffy coat instead of whole blood, laboratories gain higher cell density, clearer interfaces, and more reliable isolation outcomes, especially when isolating PBMCs or rare immune cell populations.
Buffy coat has several defining features that make it especially valuable for laboratory cell isolation workflows. These characteristics explain why it is widely used in research, diagnostics, and applied cell separation.
Highly enriched leukocyte content
Buffy coat contains leukocytes concentrated approximately 10 to 20 times higher than in whole blood. This enrichment allows researchers to start with a higher number of target cells in a smaller volume.
Reduced red blood cell background
Most erythrocytes are removed during centrifugation or filtration. This reduction simplifies downstream processing and improves clarity during density-based separations.
Lower platelet burden than whole blood
Compared to fresh whole blood, buffy coat generally contains fewer platelets, reducing interference during cell culture, flow cytometry, and enrichment steps.
Physically separated without polymers
Buffy coat preparation relies on centrifugation or filtration, not polymer-based solutions. This helps preserve native cell properties and avoids chemical exposure.
Stable and well-defined interphase
The leukocyte layer forms a visible, collectable interphase. This makes handling easier and improves consistency during sample recovery.
Ideal starting material for density centrifugation
Because cell populations are already partially enriched, buffy coat works efficiently with standard density gradient media for PBMC or leukocyte isolation.
Suitable for rare cell isolation
The higher leukocyte density improves the chances of recovering low-abundance populations, such as specific immune subsets.
Efficient use of sample volume
Concentrated cells reduce the need for large starting volumes, saving reagents, time, and processing capacity.
Compatible with multiple isolation technologies
Buffy coat integrates well with bead-based, size-based, and density-based separation methods.
Together, these characteristics make buffy coat a practical, high-value starting material for laboratories focused on reliable and scalable cell isolation workflows.
When choosing a starting material for cell isolation, laboratories often decide between whole blood and buffy coat. While both can be used successfully, they differ significantly in concentration, handling requirements, and overall efficiency.
Whole blood contains all blood components in their native proportions, red blood cells, plasma, platelets, and leukocytes. Because leukocytes represent only a small fraction of whole blood, cell isolation workflows typically require larger volumes, longer centrifugation steps, or additional pre-processing to achieve sufficient cell numbers. This can increase handling time and introduce variability, especially when working with limited samples or rare cell populations.
Buffy coat, in contrast, is already enriched for leukocytes. During blood processing, most red blood cells and plasma are removed, leaving behind a concentrated leukocyte fraction. This higher cell density allows laboratories to start with smaller volumes while still achieving strong yields. As a result, downstream steps such as density centrifugation, washing, or enrichment can be performed more efficiently.
Another important difference is background interference. Whole blood contains large numbers of erythrocytes and platelets that can complicate separation and analysis. Buffy coat reduces this background, making it easier to identify, isolate, and culture target cells.
From a workflow perspective, buffy coat often shortens processing time, reduces reagent use, and improves reproducibility. Whole blood remains valuable when fresh, minimally processed samples are required, but buffy coat is generally preferred when efficiency, yield, and consistency are priorities.
For many routine cell isolation workflows, buffy coat provides a more practical and reliable starting point than whole blood.
In addition to using buffy coat obtained from blood donation centers, laboratories can prepare their own buffy coat fraction from fresh whole blood. This approach is useful when working with small sample volumes, time-sensitive experiments, or customized workflows.
Laboratory preparation of buffy coat relies on simple, physical separation rather than chemical treatment. The goal is to gently concentrate leukocytes while keeping cells viable and functional.
A commonly used laboratory protocol includes the following steps:
Dilution of whole blood
Whole blood is mixed with an equal volume of washing buffer. Dilution reduces viscosity and supports smoother separation during centrifugation.
Low-speed centrifugation
The diluted blood is centrifuged for approximately 10 minutes at around 200 × g with the brake turned off. The low force prevents disruption of cell layers and protects fragile leukocytes.
Layer formation
After centrifugation, the sample separates into three visible layers:
Plasma at the top
A thin, cloudy interphase containing leukocytes (the buffy coat)
Red blood cells at the bottom
Buffy coat collection
The leukocyte-rich interphase is carefully removed using a pipette and transferred into a fresh tube for further processing.
This method produces a buffy coat fraction enriched in leukocytes without exposing cells to harsh conditions or polymer solutions. Because the preparation is gentle, cells remain suitable for downstream applications such as density centrifugation, enrichment, culture, or functional assays.
Preparing a buffy coat in the laboratory gives researchers flexibility and control. It allows immediate processing, reduces dependence on external sources, and supports consistent cell isolation workflows when fresh samples are required.
Buffy coat is valued in laboratory workflows because it contains a concentrated mix of immune cells that are otherwise diluted in whole blood. During separation, leukocytes accumulate in the thin interphase between plasma and red blood cells, creating a fraction that is highly suitable for cell isolation and enrichment.
The primary cell types enriched in buffy coat include:
Monocytes
These cells play a key role in immune defense and inflammation. Buffy coat provides a strong starting material for monocyte isolation, differentiation studies, and functional assays.
Lymphocytes
T cells, B cells, and natural killer (NK) cells are all present in buffy coat. Their higher concentration simplifies downstream separation and improves recovery compared to whole blood.
Peripheral Blood Mononuclear Cells (PBMCs)
Buffy coat is commonly used as a source of PBMCs, which include lymphocytes and monocytes. This makes it ideal for immunology research, cell culture, and stimulation assays.
Dendritic Cell Precursors
Although present in smaller numbers, precursors to dendritic cells can be enriched from buffy coat and used in differentiation workflows.
Because leukocytes are concentrated 10 to 20 times compared to whole blood, buffy coat allows laboratories to isolate meaningful cell numbers quickly. This enrichment reduces processing time, minimizes sample handling, and supports efficient workflows for both routine and advanced cell isolation applications.
Although a buffy coat is an excellent starting material for cell isolation, it also presents specific challenges that laboratories must manage carefully. Its high leukocyte concentration, while beneficial, can complicate handling and downstream processing if not addressed properly.
One common challenge is increased viscosity. Compared to whole blood, buffy coat is thicker and more cell-dense, which can make pipetting less precise and increase the risk of cell clumping. Without gentle handling, this can lead to uneven cell recovery or loss of fragile populations.
Platelet contamination is another frequent issue. Buffy coat often contains residual platelets that can interfere with cell culture, activation assays, and flow cytometry. Platelets may bind to leukocytes or release growth factors, altering experimental outcomes if not removed effectively.
Heterogeneous cell composition can also complicate workflows. Buffy coat contains a mix of immune cell types, requiring well-defined separation strategies to isolate specific populations with high purity.
Finally, handling-related variability can affect reproducibility. Differences in preparation methods, centrifugation settings, or storage time can influence cell viability and yield. Older buffy coat samples, in particular, may show reduced performance if not processed using optimized protocols.
Addressing these challenges requires gentle preparation, controlled separation steps, and tools designed to work with concentrated leukocyte fractions.
Buffy coat plays a central role in modern cell isolation workflows by offering a concentrated and practical source of leukocytes. Compared to whole blood, it provides higher cell numbers, reduced processing time, and greater efficiency when isolating specific immune cell populations. These advantages make buffy coat an attractive starting material for both research and applied laboratory settings.
At the same time, working with buffy coat requires an understanding of its unique properties. Its high cell density, mixed composition, and potential platelet contamination can introduce challenges if workflows are not carefully designed. Gentle handling, appropriate separation strategies, and reliable preparation methods are essential to maintain cell viability, purity, and reproducibility.
When processed correctly, buffy coat supports a wide range of applications, from basic immune research to advanced cell enrichment and downstream functional assays. Its compatibility with density-based separation and targeted isolation approaches allows laboratories to extract meaningful data while minimizing sample waste.
For laboratories seeking efficient and scalable cell isolation, buffy coat represents a valuable balance between accessibility and performance. By applying optimized protocols and choosing tools designed for concentrated leukocyte fractions, researchers can unlock the full potential of buffy coat and build workflows that are both reliable and adaptable to evolving experimental needs.